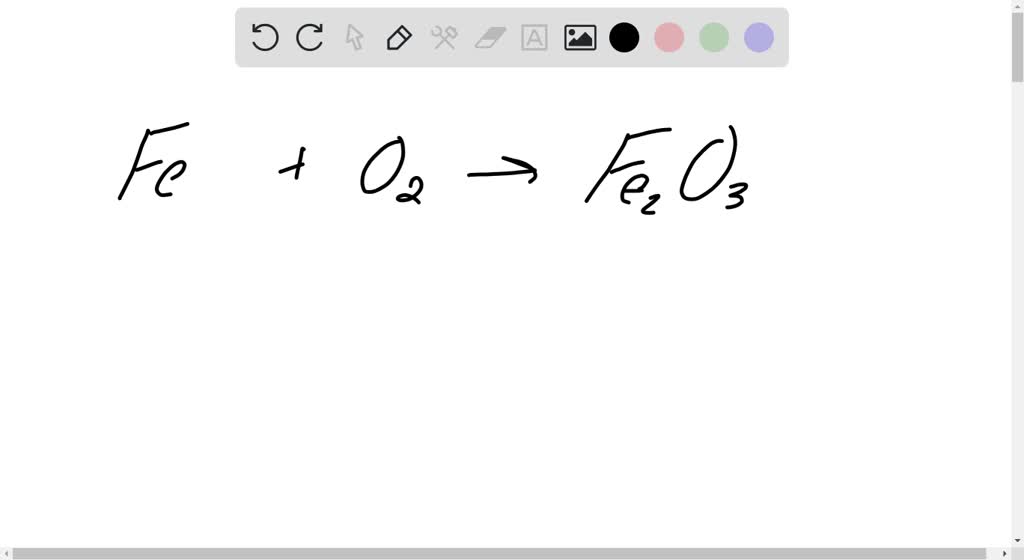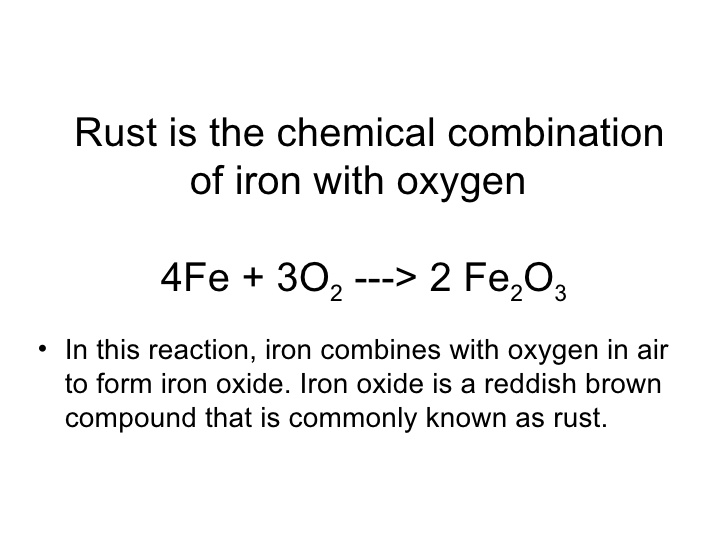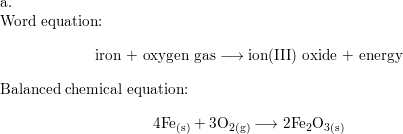Vinegar And Clr Are My New Best Friends - Iron Rusting Chemical Equation PNG Image | Transparent PNG Free Download on SeekPNG

⚗️Iron (Fe) reacts with oxygen gas (O2) to form rust (Fe2O3). Balance the equation below by writing - Brainly.com
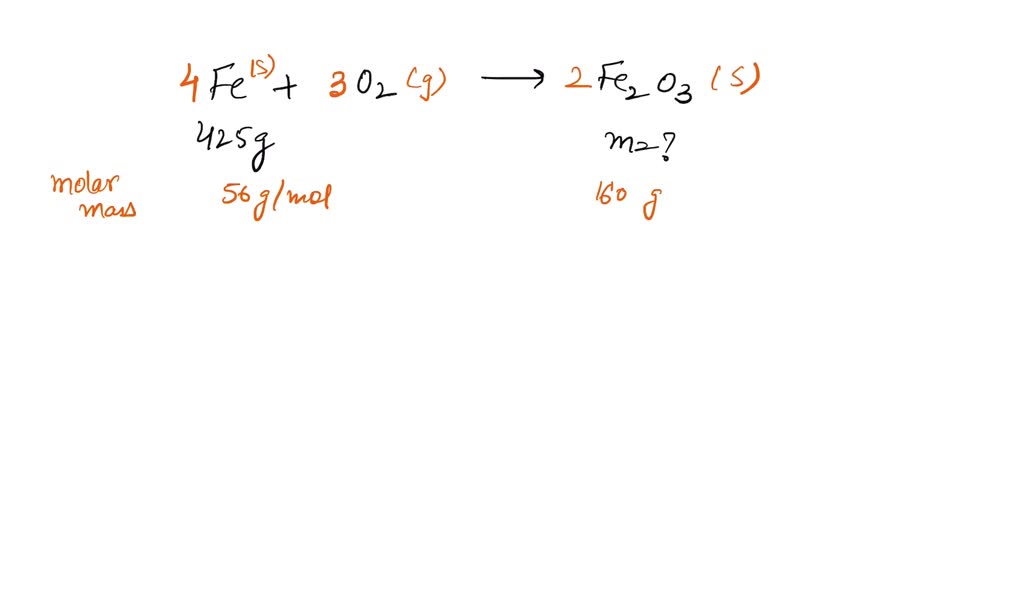
SOLVED: The formation of rust which is iron (III) oxide on the surface of iron metal is an oxidation-reduction reaction between iron metal and oxygen gas. balanced equation : Fe (s) +

What is oxidation of iron in inorganic chemistry? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
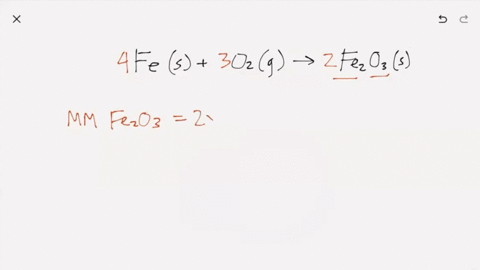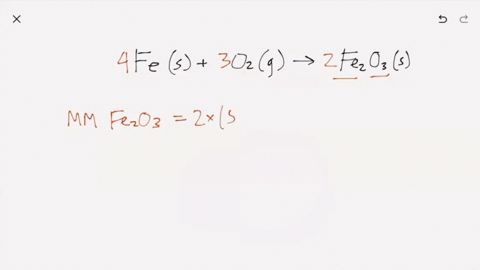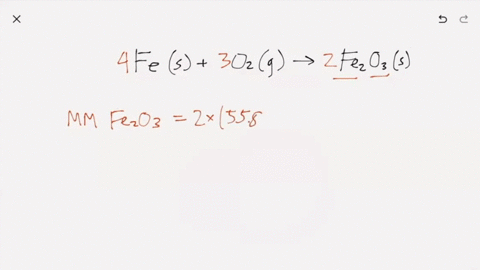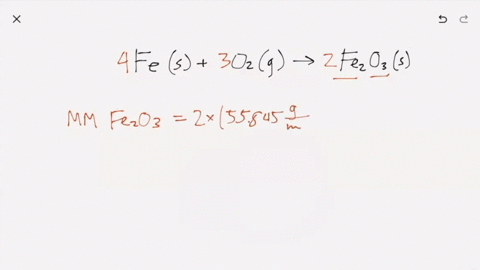
SOLVED:Iron metal reacts with oxygen to give iron(III) oxide, Fe2 O3 (a) Write a balanced equation for the reaction. (b) If an ordinary iron nail (assumed to be pure iron) has a

What does it mean for an equation to be balanced? We have the same number of each type of atom on each side of the equation. Make sure you have balanced. -